Regulatory
The Regulatory API provides access to a single comprehensive source for global Drugs & Biologics regulatory information including current and historical curated regulatory documents from authorities (covering 80 modules for country specific regulations and comparisons), summaries and reports from our experts.
Regulatory API can help you to
- See the full picture of the regulatory landscape and reach critical decisions more efficiently
- Identify common/divergent requirements across countries
- Plan your multi or single-country filings
- Stay ahead of legislations and guidelines and save time tracking regulatory changes
- Use product information tables to compare existing and emerging products
- Identify new indications for your products and prepare for committee meetings and inspections
- Guide you through individual country registration processes
- Decide the most efficient submission routes for your products
Content
- Global regulatory comparisons – Granular comparisons of regulatory requirements in countries and markets of your interest, covering the critical areas of regulatory for Drugs & Biologics
- Regulatory intelligence reports – Exclusive intelligence reports to support you with the analysis of regulatory information
- Regulatory summaries – Continuously updated explanatory documents in English providing expertise on local regulatory practices, including links and detailed summaries of reference documents
- Reference documents – Updated daily, this repository of official documents give you a complete history of the regulatory landscape, giving you additional insights and more context for your research. This also includes documents obtained through FOI requests, exclusive English translations, and historical regulatory documents that have been removed from agencies’ websites. We also include product approval information from various countries to help you compare and refine your tactical plans
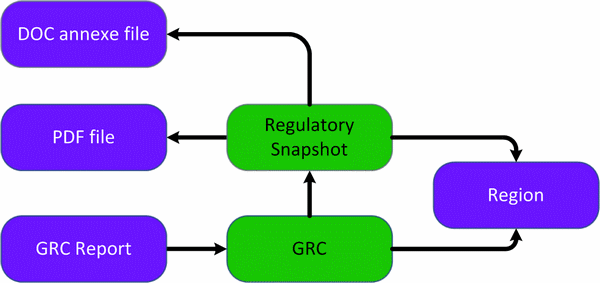
Entities diagram (Colour coding – green: main entities | purple: supporting entities)
Search results, retrieve and export regulatory documents; Retrieve and export global regulatory comparisons.